DNA Manufacturing Market (2026 – 2033)
DNA Manufacturing Market Summary
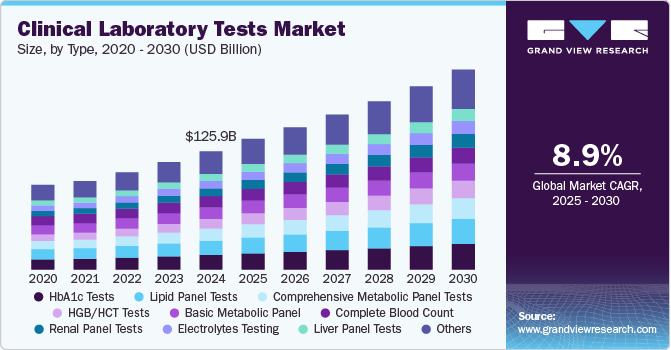
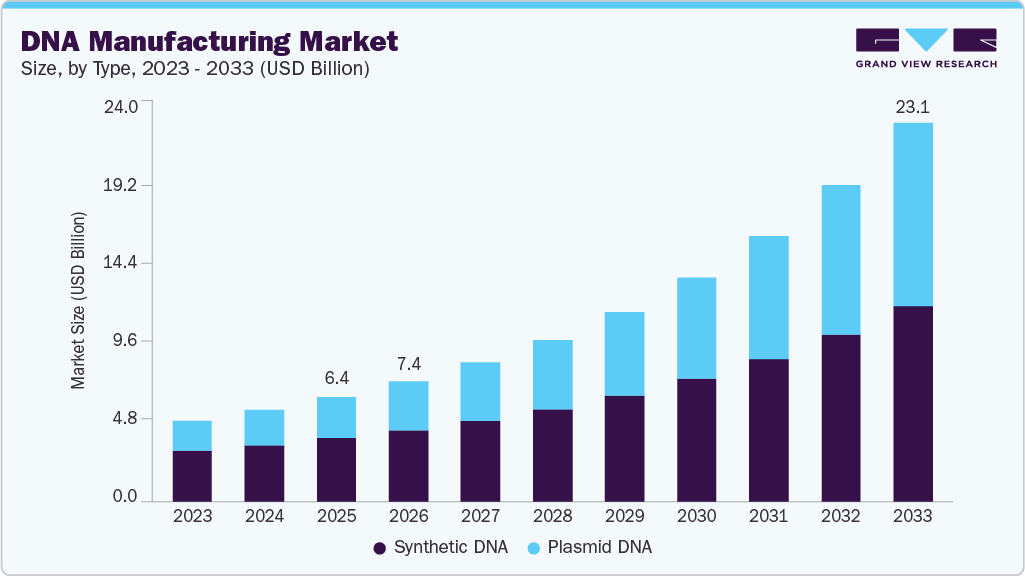
The global DNA manufacturing market size was estimated at USD 6.4 billion in 2025 and is anticipated to reach USD 23.1 billion by 2033, growing at a CAGR of 17.8% from 2026 to 2033. This growth is driven by increasing demand for gene therapies, personalized medicine, and advancements in synthetic biology.
Key Market Trends & Insights
- The North America DNA manufacturing market held the largest share of 44.5% of the global market in 2025.
- The industry in the U.S. is expected to grow significantly over the forecast period.
- By type, the synthetic DNA segment held the highest market share in 2025.
- Based on grade, the GMP grade segment held the highest market share in 2025.
- By application, the cell and gene therapy segment held the highest market share of 44.1% in 2025.
Market Size & Forecast
- 2025 Market Size: USD 6.4 Billion
- 2033 Projected Market Size: USD 23.1 Billion
- CAGR (2026-2033): 17.8%
- North America: Largest market in 2025
- Asia Pacific: Fastest growing market
Expanding applications in pharmaceutical research and vaccine development also contribute to market expansion. For instance, in July 2024, GenScript Biotech announced the launch of an advanced DNA synthesis platform that reduces production time, enabling faster development of gene therapies and synthetic vaccines.
Growth of Gene Therapy
Gene therapies are transforming the treatment landscape for genetic disorders, rare diseases, and common chronic conditions. Gene therapies directly modify or replace faulty genes within a patient’s cells, offering the potential for long-lasting or curative effects. For instance, in September 2022, GenScript USA Inc. introduced the GenWand Double-Stranded DNA (dsDNA) service, designed to enhance CRISPR-based gene editing in T-cell engineering. This service offers covalently closed, linear DNA templates that are more stable and less prone to endonuclease degradation, thereby improving homology-directed repair (HDR) efficiency and reducing off-target effects compared to traditional PCR-based methods. This innovative approach requires large-scale production of highly precise, pure DNA sequences to develop therapeutic vectors, such as viral carriers or plasmids, that deliver corrective genes into patient cells.
Recent approved gene therapies
|
Product name |
Generic name |
Year first approved |
Disease(s) |
Locations approved |
Company |
|
zevorcabtagene autoleucel |
zevorcabtagene autoleucel |
2024 |
Relapsed or refractory multiple myeloma |
China |
CARsgen Therapeutics |
|
Beqvez |
fidanacogene elaparvovec |
2024 |
Hemophilia B |
Canada |
Pfizer |
|
Lyfgenia |
lovotibeglogene autotemcel |
2023 |
Sickle cell anemia |
U.S. |
bluebird bio |
|
inaticabtagene autoleucell |
inaticabtagene autoleuce |
2023 |
Acute lymphocytic leukemia |
China |
Juventas Cell Therapy |
|
Casgevy |
exagamglogene autotemcel |
2023 |
Sickle cell anemia; thalassemia |
U.S., UK, Bahrain, Saudi Arabia, EU |
CRISPR Therapeutics |
|
Fucaso |
equecabtagene autoleucel |
2023 |
Multiple myeloma |
China |
Nanjing IASO Biotechnology |
|
Vyjuvek |
beremagene geperpavec |
2023 |
Dystrophic epidermolysis bullosa |
U.S. |
Krystal Biotech |
|
Elevidys |
delandistrogene moxeparvovec |
2023 |
Duchenne muscular dystrophy |
U.S. |
Sarepta Therapeutics |